Metamorphosis of whitemouth croaker Micropogonias furnieri (Pisces, Sciaenidae)
DOI:
https://doi.org/10.47193/mafis.3322020301107Keywords:
Sciaenidae, larvae transformation, ontogeny, morphology, morphometryAbstract
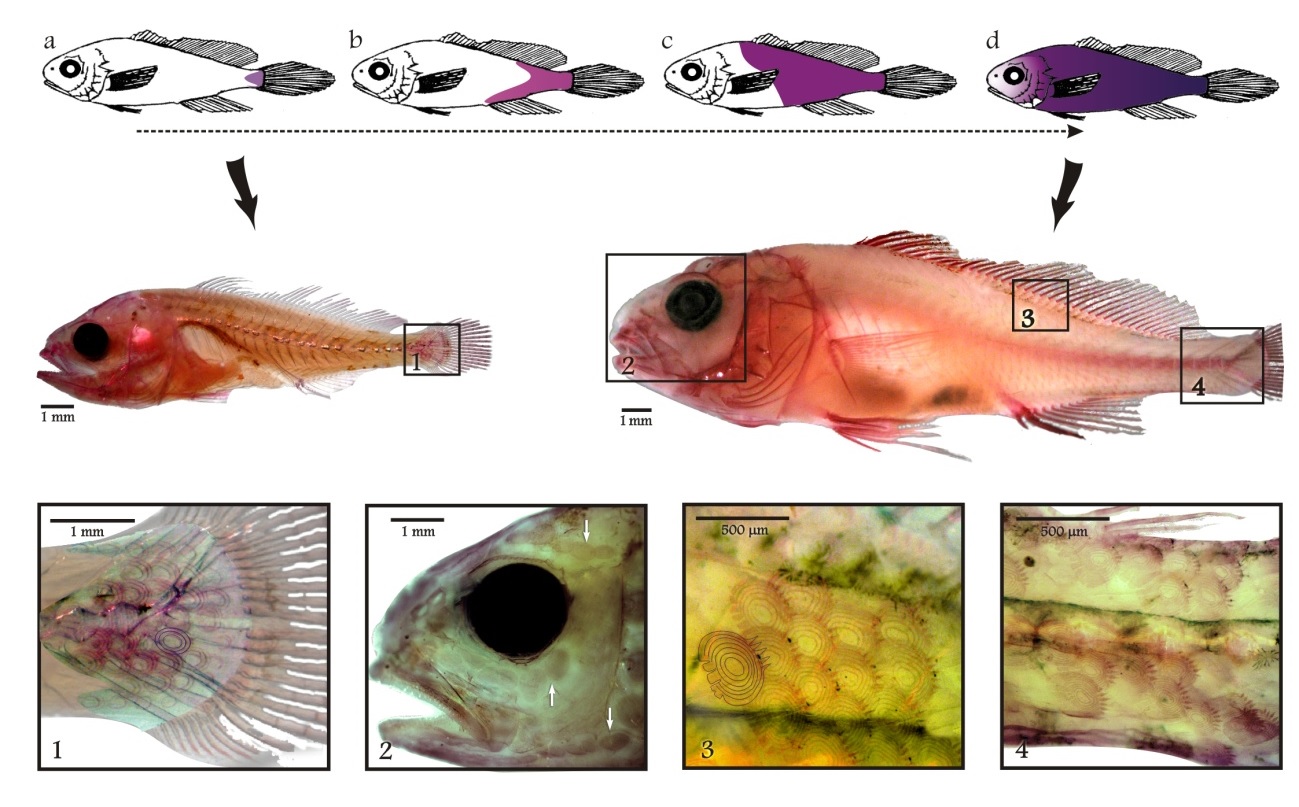
Fish metamorphosis is an important ontogenetic process with a key role on early stages survival and on successful recruitment to adult populations. The whitemouth croaker (Micropogonias furnieri) is an important commercial resource for the coastal fisheries of Argentina and Uruguay. Metamorphosis was studied using morphometric and morphological analysis during larval development. Changes in morpho-meristic characters before and after metamorphosis were employed to determine the length interval of this transition by employing Principal Component Analysis. Individuals (n = 430) from 4 to 41 mm standard length (SL) were collected in the Río de la Plata estuary (35.45° S, 56.35° W) in March 2006. Length ranges of individual’s developmental stages were associated with the presence of key morphological characters. During early life stages, M. furnieri changes from a big-headed, robust shape larva to a slender and more elongated body form. Most of the morphometric variables showed an inflexion point at 15.2 mm SL, with a 95% confidence interval of 14.0-16.4 mm. The anterior part of the body grows faster during early stages, probably related to an intense feeding activity strategy. The completion of pectoral fin rays and the onset of squamation determine the beginning of metamorphosis at 11-12 mm SL. At around 18 mm SL, squamation ends, first barbels develop and the sagittae otoliths primordium is closed. The length-at-metamorphosis for M. furnieri was established between 9 to 18 mm SL, since all developmental characters studied highly overlapped at that interval. All those processes are indicative of the beginning of the juvenile period associated to the settlement and the start of a bottom-oriented life-style.
Downloads
References
Acha EM, Mianzan HW, Guerrero RA, Carreto J, Giberto D, Montoya N, Carignan M. 2008. An overview of physical and ecological processes in the Río de la Plata Estuary. Cont Shelf Res. 28: 1579-1588.
Acha EM, Mianzan HW, Lasta CA, Guerrero RA. 1999. Estuarine spawning of the whitemouth croaker Micropogonias furnieri in the Río de la Plata, Argentina. Mar Freshwater Res. 50: 57-65.
Balon EK. 1984. Reflections on some decisive events in the early life of fishes. Trans Am Fish Soc. 113: 178-185.
Balon EK. 1989. The Tao of life: from the dynamic unity of polar opposites to self-organization. In: Bruton MN, editor. Alternative life-history styles of animals. Perspectives in vertebrate science. Vol. 6. Dordrecht: Kluwer Academic Publishers. p. 7-40.
Boehlert GW, Mundy BC. 1988. Roles of behavioral and physical factors in larval and juveniles fish recruitment to estuarine nursery areas. Am Fish Soc Symp. 3: 51-67.
Boglione C, Giganti M, Selmo C, Cataudella S. 2003. Morphoecology in larval fin-fish: a new candidate species for aquaculture, Diplodus puntazzo (Sparidae). Aquacult Int. 11: 17-41.
Braverman MS, Acha EM, Gagliardini DA, Rivarossa M. 2009. Distribution of whitemouth croaker (Micropogonias furnieri, Desmarest 1823) larvae in the Río de la Plata estuarine front. Estuar Coast Shelf Sci. 82: 557-565.
Braverman M, Brown DR, Acha EM. 2015. Indirect validation of daily increments in whitemouth croaker (Micropogonias furnieri) larvae otoliths. Rev Invest Desarr Pesq. 26: 59-67.
Buratti CC, Santos BA. 2010. Otolith microstructure and pelagic larval duration in two stocks of the Argentine hake, Merluccius hubbsi. Fish Res. 106: 2-7.
Campana SE, Nielson JD. 1985. Microstructure of fish otoliths. Can J Fish Aquat Sci. 42: 1014-1032.
Carozza CR, Lasta CA, Ruarte C, Cotrina CP, Mianzan H, Acha EM. 2004. Corvina rubia (Micropogonias furnieri). In: Sánchez RP, Bezzi SI, editors. El Mar Argentino y sus recursos pesqueros. Tomo 4. Los peces marinos de interés pesquero. Caracterización biológica y evaluación del estado de explotación. Mar del Plata: Instituto Nacional de Investigación y Desarrollo Pesquero (INIDEP). p. 255-270.
Copp GH, Kováč V. 1996. When do fish with indirect development become juveniles? Can J Fish Aquat Sci. 53: 746-752.
Dando PR. 1984. Reproduction in estuarine fish. In: Potts GW, Wootton RJ, editors. Fish reproduction: strategies and tactics. London: Academic Press. p. 155-170.
Ditty JG, Fuiman LA, Shaw RF. 2003. Characterizing natural intervals of development in fishes: an example using blennies (Teleostei: Blenniidae). In: The big fish bang. Proceedings of the 26th annual larval fish conference. Bergen. p. 405-418.
Fuiman LA. 1983. Growth gradients in fish larvae. J. Fish Biol. 23: 117-123.
Fuiman LA, Poling KR, Higgs DM. 1998. Quantifying developmental progress for comparative studies of larval fishes. Copeia. 3: 602-611.
Fuiman LA, Werner RG. 2002. Fishery science: the unique contributions of early life stages. Oxford: Blackwell Science Ltd. Osney Mead. 326 p.
Govoni JJ. 2004. The development of form and function in fishes and the question of larval adaptation. Am Fish Soc Symp. 40: 1-7.
Gozlan RE, Copp GH, Tourenq JN. 1999. Comparison of growth plasticity in the laboratory and field, and implications for the onset of juvenile development in sofie, Chondrostoma toxostoma. Environ Biol Fish. 56: 153-165.
Haedrich RL. 1992. Estuarine fishes. In: Ketchum BH, editor. Ecosystems of the world. Estuaries and enclosed seas. Vol 26. Amsterdam: Elsevier. p. 185-207.
Huxley J. 1932. Problems of relative growth. New York: New York Dial Press.
Jolicoeur P. 1963a. The multivariate generalization of the allometry equation. Biometrics. 19: 497-499.
Jolicoeur P. 1963b. The degree of generality of robustness in Martes americana. Growth. 27: 1-27.
Juanes F, Conover DO. 1994. Rapid growth, high feeding rates, and early piscivory in young-of-the-year bluefish (Pomatomus saltatrix). Can J Fish Aquat Sci. 51: 1752-1761.
Kanou K, Kohno H, Sano M. 2004. Morphological and functional development of characters associated with settlement in the yellowfin goby, Acanthogobius flavimanus. Ichthyol Res. 51: 213-221.
Kolkovski S, Tandler A, Kissil GW, Gertler A. 1993. The effect of dietary exogenous digestive enzymes on ingestion, assimilation, growth and survival of gilthead seabream (Sparus aurata, Sparidae, Linnaeus) larvae. Fish Physiol Biochem. 12: 203-209.
Koumoundouros G, Ashton C, Xenikoudakis G, Giopanou I, Georgakopoulou E, Stickland N. 2009. Ontogenetic differentiation of swimming performance in Gilthead seabream (Sparus aurata, Linnaeus 1758) during metamorphosis. J Exp Mar Biol Ecol. 370: 75-81.
Kováč V. 2002. Synchrony and heterochrony in ontogeny (of fish). J Theor Biol. 217: 499-507.
Kováč V, Copp GH. 1999. Prelude: looking at early development in fishes. Environ Biol Fish. 56: 7-14.
Kováč V, Katina S, Copp GH, Siryova S. 2006. Ontogenetic variability in external morphology and microhabitat use of spirlin Alburnoides bipunctatus from the River Rudava (Danube catchment). J Fish Biol. 68: 1257-1270.
Lauff M, Hoffer R. 1984. Proteolytic enzymes in fish development and the importance of dietary enzymes. Aquaculture. 37: 335-346.
Macchi GJ, Christiansen HE. 1996. Análisis temporal del proceso de maduración y determinación de la incidencia de atresias en la corvina rubia (Micropogonias furnieri). Frente Marit. 16: 93-101.
Mann KH, Lazier JRN. 1996. Dynamics of marine ecosystems: biological-physical interactions in the oceans. Malden: Blackwell Science. 394 p.
McCormick MI, Makey L, Dufour V. 2002. Comparative study of metamorphosis in tropical reef fishes. Mar Biol. 141: 841-853.
Mianzan H, Lasta C, Acha EM, Guerrero R, Macchi GJ, Bremec C. 2001. The Río de la Plata Estuary, Argentina-Uruguay. In: Seeliger U, Kjerfve B, editors. Ecological studies. Coastal marine ecosystems of Latin America. Berlin: Springer-Verlag. p. 185-204.
Miller MJ, Nemerson DM, Able KW. 2003. Seasonal distribution, abundance, and growth of young-of-the-year Atlantic croaker (Micropogonias undulatus) in Delaware Bay and adjacent marshes. Fish Bull. 101 (1): 100-115.
Morales-Nin B, Aldebert Y. 1997. Growth of juvenile Merluccius merluccius in the Gulf of Lions (NW Mediterranean) based on otolith microstructure and length-frequency analysis. Fish Res. 30 (1-2): 77-85.
Morioka S, Machinandiarena L, Villarino MF. 2001. Preliminary information on internal structures of otoliths and growth of ling, Genypterus blacodes (Ophidiidae), larvae and juveniles collected off Argentina. Bull Japan Soc Fish Ocean. 65 (2): 59-66.
Moser HG. 1996. Principles and terminology. The early stages of fishes in the California current region. California cooperative oceanic fisheries investigations. Atlas. 33. p. 27-44.
Nikolioudakis N, Koumoundouros G, Kiparissis S, Somarakis S. 2010. Defining length-at-metamorphosis in fishes: a multi-character approach. Mar Biol. 157: 991-1001.
Osse JWM, van den Boogaart JGM. 1995. Fish larvae, development, allometric growth and the aquatic environment. ICES Mar Sci Symp. 201: 21-34.
Osse JWM, van den Boogaart JGM, van Snik GMJ, van der Sluys L. 1997. Priorities during early growth of fish larvae. Aquaculture. 155: 249-258.
Pavlov DA. 1999. Features of transition from larva to juvenile in fishes with different types of early ontogeny. Environ Biol Fish. 56: 41-52.
Pedersen BH, Nilssen EM, Hjelmeland K. 1987. Variations in the content of trysin and trysinogen in larval herring (Clupea harengus) digesting copepod nauplii. Mar Biol. 94: 171-181.
Potthoff T. 1984. Clearing and staining techniques. In: Lawrence KS, Moser HG, Richards WJ, Cohen DM, Fahay MP, Kendall AW Jr, Richardson SL, editors. Ontogeny and systematics of fishes. Special Publication 1. American Society of Ichthyologists and Herpetologist. p. 33-37.
Rodríguez-Graña L, Vera M, Cervetto G, Calliari DL. 2018. Trophic ecology of the White Croaker (Micropogonias fur-nieri Desmarest, 1823) and Rough Scad (Trachurus lathami Nichols, 1920) larvae in the Río de la Plata Estuary. In: Hoffmeyer M, Sabatini M, Brandini F, Calliari D, Santinelli N, editors. Plankton ecology of the Southwestern Atlantic. Cham: Springer. p. 349-371.
Rooker JR, Holt SA. 1997. Utilization of subtropical seagrass meadows by newly settled red drum (Sciaenops ocellatus): patterns of distribution and growth. Mar Ecol Prog Ser. 158: 139-149.
Secor HD. 2015. Migration ecology of marine fishes. Baltimore: Johns Hopkins University Press. 292 p.
Shea BT. 1985. Bivariate and multivariate growth allometry: statistical and biological considerations. J Zool. 206: 367-390.
Simionato CG, Berasategui AD, Meccia V, Acha EM, Mianzan H. 2008. Short time-scale wind forced variability in the Río de la Plata Estuary and its role on ichthyoplankton retention. Estuar Coast Shelf Sci. 76: 211-226.
Sinque C. 1980. Larvas de Sciaenidae (Teleostei) identificadas na região estuarino-lagunar de Cananéia. Universidade do São Paulo. Bol Zool. 5: 39-77.
Smith PE. 1985. Year class strength and survival of 0-group clupeoids. Can J Fish Aquat Sci. 42: 69-82.
Sogard SM. 1991. Interpretation of otolith microstructure in juvenile winter flounder (Pseudopleuronectes americanus): ontogenetic development, daily increment validation, and somatic growth relationships. Can J Fish Aquat Sci. 48: 1862-1871.
Taylor WR, Van Dyke GC. 1985. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cibium. 9 (2): 107-119.
Urho L. 2002. Characters of larvae-what are they? Folia Zool. 51: 161-186.
Weiss G. 1981. Ictioplancton del estuario de Lagoa dos Patos, Brasil [tesis doctoral]. La Plata: Facultad de Ciencias Naturales y Museo, Universidad Nacional de La Plata. 164 p.
Werner RG. 2002. Habitat requeriments In: Fuiman LA, Werner RG, editors. Fishery Science. The unique contributions of early life stages. Oxford: Blackwell Publishing. p. 161-182.
Wilson DT, McCormick MI. 1997. Spatial and temporal validation of settlement marks in the otoliths of tropical reef fishes. Mar Ecol Prog Ser. 153: 259-271.
Wilson DT, McCormick MI. 1999. Microstructure of settlement-marks in the otoliths of tropical reef fishes. Mar Biol. 134: 29-41.
Xie S, Watanabe Y, Saruwatari T, Masuda R, Yamashita Y, Sassa C, Konishi Y. 2005. Growth and morphological development of sagittal otoliths of larval and early juvenile Trachurus japonicus. J Fish Biol. 66: 1704-1719. DOI: https://doi.org/10.1111/j.0022-1112.2005.00717.x
Published
Issue
Section
License
Copyright (c) 2020 Mara S. Braverman , Daniel Brown , E. Marcelo Acha

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Authors of articles published in Marine and Fishery Sciences retain copyright on their articles, except for any third-party images and other materials added by Marine and Fishery Sciences, which are subject to copyright of their respective owners. Authors are therefore free to disseminate and re-publish their articles, subject to any requirements of third-party copyright owners and subject to the original publication being fully cited. Visitors may also download and forward articles subject to the citation requirements. The ability to copy, download, forward or otherwise distribute any materials is always subject to any copyright notices displayed. Copyright notices must be displayed prominently and may not be obliterated, deleted or hidden, totally or partially.
This journal offers authors an Open Access policy. Users are allowed to read, download, copy, distribute, print, search, or link to the full texts of the articles, or use them for any other legal purpose within the Creative Commons 4.0 license (BY-NC-SA), without asking prior permission from the publisher or the author. This is in accordance with the BOAI definition of Open Access.