Culture of Bacillus subtilis and Saccharomyces cerevisiae from byproducts of the enzymatic extraction of Atlantoraja castelnaui oil
DOI:
https://doi.org/10.47193/mafis.3822025010104Keywords:
Cartilaginous species, fish liver, oil extraction, culture media, microbial growth, circular economyAbstract
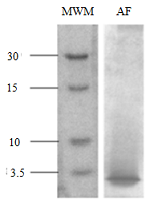
The growing trend in global fish consumption has led to the generation of a large amount of waste and byproducts with negative economic and environmental impacts. The production of fish oil from fish processing residues seems to be a sustainable and future opportunity to provide valuable fatty acids for animal and human consumption. In parallel, this alternative valorization of fish waste has increased the demand to explore sustainable extraction methods. Enzymatic hydrolysis is an efficient, rapid and reproducible method for the extraction of oils from fish guts, producing an aqueous phase rich in proteins and soluble compounds. In this work, a highly soluble aqueous phase obtained from the enzymatic extraction of the liver oil of the ray Atlantoraja castelnaui was tested as a source of nutrient for the growth of Bacillus subtilis and Saccharomyces cerevisiae. Culture media were supplemented with the aqueous phase at a final concentration of 10 mg ml-1 total protein. The growth pattern and biomass yield of yeasts cultured in yeast extract peptone-dextrose (YPD) medium did not show statistically significant differences (p: 0.05) with the diluted medium and the aqueous fraction (AF) supplemented medium. Similar results were obtained for B. subtillis and its positive control in Luria Bertani (LB) medium. In both cases, the ability of these inexpensive media to support the growth of microorganisms was demonstrated. Results suggested that the remaining aqueous phase of the discards from A. castelnaui oil production can be used as an alternative substrate for microorganism culture purposes. In this way, a residue destined for disposal could become a product with added value achieving a good result in the context of circular economy.
Downloads
References
Aldedayo MR, Ajiboye EA, Akintunde JK, Odaibo A. 2014. Single cell proteins: as nutritional enhancer. Adv Appl Sci Res. 2: 396-409.
Aggelopoulos T, Katsieris K, Bekatorou A, Pandey A, Banat IM, Koutinas AA. 2014. Solid state fermentation of food waste mixtures for single cell protein, aroma volatiles and fat production. Food Chem. 145: 710-716.
Aspmo SI, Horn SJ, Eijsink VGH. 2005. Use of hydrolysates from Atlantic cod (Gadus morhua L.) viscera as a complex nitrogen source for lactic acid bacteria. FEMS Microbiol Lett. 248: 65-68.
Binupriya AR, Sathishkumar M, Sub Ku C, Yun S. 2010. Sequestration of reactive Blue 4 by free and immobilized Bacillus subtilis cells and its extracellular polysaccharides. Biointerfaces. 76: 179-185.
Czerucka D, Piche T, Rampal P. 2007. Review article: yeast as probiotics--Saccharomyces boulardii. Aliment Pharmaco Ther. 26 (6): 767-778.
Datta S, Timson D, Annapure US. 2017. Antioxidant properties and global metabolite screening of the probiotic yeast Saccharomyces cerevisiae var. boulardii. J Sci Food Agric. 97: 3039-3049.
Deraz SF, El-Fawal GF, Abd-Ellatif SA, Khalil AA. 2011. Autohydrolysed Tilapia nilotica fish viscera as a peptone source in bacteriocin production. Indian J Microbiol. 51: 171-175.
Duarte L, Carvalehiro F, Lopes S, Neves I, Gírio F. 2008. Yeast biomass production in brewery’s spent grains hemicellulosic hydrolyzate. Appl Biochem Biotech. 148: 119-129.
Fallah M, Baharam S, Javadiam SR. 2015. Fish peptone development using enzymatic hydrolysis of silver carp by-products as a nitrogen source in Staphylococcus aureus media. Food Sci Nutr. 3: 153-157.
[FDA] U.S. Food and Drugs Administration. 2018. Microorganisms & microbial-derived ingredients used in food (partial list). [accessed 2021 Mar 26]. Silver Spring: FDA. https://www.fda.gov/food/generally-recognized-safe-gras/microorganisms-microbial-derived-ingredients-used-food-partial-list.
Fernandes PL, Rodrigues EM, Paiva FR, Ayupe BAL, McInerney MJ, Tótola MR. 2016. Biosurfactant, solvents and polymer production by Bacillus subtilis RI4914 and their application for enhanced oil recovery. Fuel. 180: 551-557.
Garimelas S, Kundle KR, Kasoju A, Meregu R. 2017. Current status on Single Cell Protein (SCP) production from photosynthetic purple non-sulphur bacteria. J Pharm Health Care Sci. 10: 915-922.
Ghaly A, Ramakrishnan V, Brooks M, Budge S, Dave D. 2013. Fish processing wastes as a potential source of proteins, amino acids and oils: a critical review. Microb Biochem Technol. 5: 4.
Gomashe AV, Pounikar MA, Gulhane PA. 2010. Liquid whey, a potential substrate for single cell protein production from Bacillus subtilis. Int J Life Sciences. 2: 119-123.
Jabeen S, Khan MAS, Hassa QM, Ahmed MS, Nishat N, Zain H. 2015. Comparative study of bioremediation of crude oil by Bacillus subtilis and organic substances. JCBPS Sec B. 5 (3): 2621-2633.
Jiaqian W, Jinlong H, Shuminao Z, Mingxion H, Guoquan H, Xiangyang G, Nan P. 2018. Single-cell protein and xylitol production by a novel yeast strain Candida intermedia FL023 from kignocellulosic hydrolysates and xylose. Appl Biochem Biotechnol. 185: 163-178
Kogje A, Ghosalkar A. 2016. Xylitol production by Saccharomyces cerevisiae overexpressing different xylose reductases using non-detoxified hemicellulosic hydrolysate of corncob. 3 Biotech. 6 (2): 127.
Kurbanoglu EK, Algur OF. 2002. Single-cell protein production from ram horn hydrolysate by bacteria. Bioresour Technol. 85: 125-129.
Laemli UK. 1970. Cleavage of structructural proteins during assembly of the head of bacteriophage T4. Nature. 227: 680-685.
Lamas DL, Massa AE. 2019. Ray liver oils obtained by different methodologies: characterization and refining. J Aquat Food Prod Technol. 28: 555-569.
Lamas DL, Massa AE. 2022. Hidrólisis enzimática de residuos del procesamiento de surel (Trachurus lathami): caracterización de las fracciones obtenidas. Revista Científica. 30 (2).
Lamas DL, Massa AE. 2023. Extraction and purification of enzymes from the Southern eagle ray (Myliobatis goodei) by-products and their compatibility with detergents: a practical approach towards circular economy. Iran J Fish Sci. 22 (4): 853-870.
Lamas DL, Massa AE. 2024. Narrownose smoothhound (Mustelus schmitti) shark liver: from a residue to a high added value biocompounds. Food Chem Adv. 4: 100623.
Martone C, Pérez Borla O, Sánchez J. 2005. Fishery by-product as a nutrient source for bacteria and archaea growth media. Bioresource Technol. 96: 383-387.
Melnykov AV. 2016. New mechanisms that regulate Saccharomyces cerevisiae short peptide transporter achieve balanced intracellular amino acid concentrations. Yeast. 33: 21-31.
Mercado Flores Y, Cardenas Alvares IO, Rojas Olvra AV, Perez Camarillo JP, Leyva MIR SG, Anducho Reyes MA. 2014. Application of Bacillus subtilis in the biological control of the phytopathogenic fungus Sporisorium reilianum. Biol Control. 76: 36-40.
Mirzapour-Kouhdasht A, Moosavi-Nasab M, Yousefi R, Eun JB. 2021. Bio/Multi-functional peptides derived from fish gelatin hydrolysates: technological and functional properties. Biocatal Agric Biotechnol. 36: 102152.
Mozumder MMH, Uddin MM, Schneider P, Raiyan MHI, Trisha MGA, Tahsin TH, Newase S. 2022. Sustainable utilization of fishery waste in Bangladesh-a qualitative study for a circular bioeconomy initiative. Fishes. 7: 84. DOI: https://doi.org/10.3390/fishes7020084
Mukhin VA, Novikov YU V, Ryzhikova LS. 2001. A protein hydrolysate enzymatically produced from the industrial waste of processing icelandic scallop Chlamys islandica. Appl Biochem Microbiol. 37: 292-296.
Nawaz A, Li, E, Irshad S, Xiong Z, Xiong H, Shahbaz HM, Siddique F. 2020. Valorization of fisheries by-products: challenges and technical concerns to food industry. Trends Food Sci Technol. 99: 34-43.
Paulo JA, O’Connell JD, Gaun A, Dydi ST. 2015. Proteome-wide quantitative multiplexed profiling of protein expression: carbon-source dependency in Saccharomyces cerevisiae. Mol Biol Cell. 26 (22): 4063-4074.
Petrova I, Tolstorebrov I, Zhivlyantseva I, Eikevik TM. 2021. Utilization of fish protein hydrolysates as peptones for microbiological culture medias. Food Biosci. 42: 101063.
Poernomo A, Buckle KA. 2002. Crude peptones from cowtail ray (Trygon sephen) viscera as microbial growth media. World J Microbiol Biotechnol. 18: 337-344.
Ramkumar A, Sivakumar N, Victor R. 2016. Fish waste-potential low cost substrate for bacterial protease production: a brief review. Open Biotechnol J. 10: 335-341.
Rebah FB, Miled N. 2013. Fish processing wastes for microbial enzyme production: a review. Biotech. 3: 255-265.
Safari R, Motamedzadegan A, Ovissipour M, Regenstein JM, Gildberg A, Rasco B. 2012. Use of hydrolysates from yellowfin tuna (Thunnus albacares) heads as a complex nitrogen source for lactic acid bacteria. Food Bioprocess Technol. 5: 73-79.
Salomone AL, Lamas DL. 2024. Valorization of the discards from the extraction oil processes of a ray fish (Zearaja flavirostris) liver as a culture media to produce Saccharomyces cerevisiae biomass. Iran J Fish Sci. 23 (2): 337-348.
Schultz N, Chang L, Hauck A, Reuss M, Syldatk C. 2006. Microbial production of single-cell protein from deproteinized whey concentrates. Appl Microbiol Biotechnol. 69: 515-520.
Shi S, Li J, Blersch DM. 2018. Utilization of solid catfish manure waste as carbon and nutrient source for lactic acid production. Appl Microbiol Biotechnol. 102: 4765-4772.
Soto Deza NM, Lopez Medina SE, Murguia Reyes CS. 2012. Eficacia de la cepa nativa de Bacillus subtilis como agente supresor del nematodo del nudo Meloidogyne spp. en cultivo de Capsicum annuum (ají pimiento piquillo). Rev Inv Agr Amb. 3: 25-40.
Triki-Ellouz Y, Ghorbel B, Souissi N, Kammoun S, Nasri M. 2003. Biosynthesis of protease by Pseudomonas aeruginosa MN7 grown on fish substrate. World J Microbiol Biotechnol. 19: 41-45.
Turcotte B, Liang XB, Robert F, Soontoyngun N. 2010. Transcriptional regulation of nonfermentable carbon utilization in budding yeast. FEMS Yeast Res. 10: 2-13.
Vázquez JA, Durán AI, Menduíña A, Nogueira M. 2020. Biotechnological valorization of food marine wastes: microbial productions on peptones obtained from aquaculture by-products. Biomolecules. 10 (8): 1184.
Vázquez JA, Pedreira A, Salmerón I, Wardhani DH, Valcarcel J. 2023. The fermentation of a marine probiotic bacterium on low-cost media formulated with industrial fish gelatin waterstreams and collagen hydrolysates. Processes. 11 (8): 2397. DOI: https://doi.org/10.3390/pr11082397
Yalçm S, Yalçm S, Can P, Gürdal AO, Bagci C, Eltan O. 2011. The nutritive value of live yeast culture (Saccharomyces cerevisiae) and its effect on milk yield, milk composition and some blood parameters of dairy cows. Asian Aust J Anim Sci. 24 (10): 1377-1385.
Published
Issue
Section
License
Copyright (c) 2024 Andrea L. Salomone †, Daniela L. Lamas

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Authors of articles published in Marine and Fishery Sciences retain copyright on their articles, except for any third-party images and other materials added by Marine and Fishery Sciences, which are subject to copyright of their respective owners. Authors are therefore free to disseminate and re-publish their articles, subject to any requirements of third-party copyright owners and subject to the original publication being fully cited. Visitors may also download and forward articles subject to the citation requirements. The ability to copy, download, forward or otherwise distribute any materials is always subject to any copyright notices displayed. Copyright notices must be displayed prominently and may not be obliterated, deleted or hidden, totally or partially.
This journal offers authors an Open Access policy. Users are allowed to read, download, copy, distribute, print, search, or link to the full texts of the articles, or use them for any other legal purpose within the Creative Commons 4.0 license (BY-NC-SA), without asking prior permission from the publisher or the author. This is in accordance with the BOAI definition of Open Access.