Efectos de cantaxantina dietaria sobre el estres por radiacion ultravioleta en el camarón Artemesia longinaris
DOI:
https://doi.org/10.47193/mafis.3632023010904Palabras clave:
Crustáceos, carotenoides, fotoprotección, actividad antioxidanteResumen
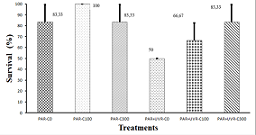
Los objetivos de esta investigación fueron investigar los efectos de dietas adicionadas con cataxantina sintética (10% parafarm) y evaluar su posible papel protector bajo la radiación ultravioleta (RUV) en el camarón Artemesia longinaris. Se prepararon tres dietas isoprotéicas e isolipídicas (41% proteína y 12% lípidos) con 0 (C0), 100 (C100) y 300 (C300) mg de cantaxantina kg-1 de dieta. Previo al experimento de radiación, los camarones fueron alimentados con las diferentes dietas durante 21 d para determinar una posible acumulación de carotenoides. Posteriormente, los animales fueron expuestos a dos tratamientos de radiación durante 7 d: a) radiación fotosintéticamente activa (PAR, 400-700 nm), y b) radiación total (PAR+RUV, 280-700 nm), bajo condiciones controladas (19 ± 2 °C, salinidad = 33, pH = 7). En los individuos expuestos al tratamiento PAR+RUV, la supervivencia varió entre 50 y 83,33%, con el valor más alto en animales alimentados con dieta C300. Al final del experimento, se registraron diferencias estadísticas significativas en la concentración de carotenoides en el tegumento. Bajo estrés por RUV se registró la mayor disminución de carotenoides no polares y astaxantina esterificada en camarones alimentados con dietas con cataxantina. La capacidad antioxidante se evaluó mediante espectroscopía de resonancia electrónica (EPR) utilizando el radical estable 2,2-difenil-2-picrilhidrazilo (DPPH). Los camarones alimentados con C300 mostraron la mayor actividad evidenciada por el decaimiento de DPPH. Los resultados sugirieron que la cantaxantina dietaria podría estar actuando como un antioxidante contra las especies reactivas de oxígeno y producir una alta tolerancia bajo estrés por RUV.
Descargas
Referencias
Ando S, Takeyama T, Hatano M. 1986. Isolation and characterization of a carotenoid-carrying lipoprotein in the serum of Chum salmon (Oncorhynchus keta) during spawning migration. Agric Biol Chem. 50: 907-914.
Arzoz NS, Marcoval MA, Díaz AC, Espino ML, Velurtas MS, Churio MS, Orallo DE, Fenucci JL. 2022. Photoprotection effects of red seaweed meal (Grateloupia cf. turuturu) as feed ingredient in prawn Artemesia longinaris. Photochem Photobiol. 98 (6): 1476-1485.
Bacchetta C, Rossi AS, Cian RE, Drago SR, Cazenave J. 2019. Dietary β-carotene improves growth performance and antioxidant status of juvenile Piaractus mesopotamicus. Aquacult Nutr. 25 (4): 761-769. DOI: https://doi.org/10.1111/anu.12893
Barnes PW, Williamson CE, Lucas RM, Robinson SA, Madronich S, Nigel D, Bornman JF, Bais AF, Sulzberger B, Wilson SR, et al. 2019. Ozone depletion, ultraviolet radiation, climate change and prospects for a sustainable future. Nat Sust. [accessed 2021 Apr 25]; 2: 569-579. DOI: https://doi.org/10.1038/s41893-019-0314-2
Bauer RT. 2020. Shrimp fisheries. Fish Aquacult. 9: 91-116.
Boonyaratpalin M, Thongrod S, Supamattaya K, Britton G, Schlipalius LE. 2001. Effects of beta-carotene source, Dunaliella salina, and astaxanthin on pigmentation, growth, survival and health of Penaeus monodon. Aquac Res. 32: 182-190.
Carreto JI, Carignan MO. 1984. Pigmentos carotenoides del camarón Artemesia longinaris Bate (Crustacea, Decapoda, Penaeidae). Rev Invest Desarr Pesq. 4: 5-20.
Castillo R, Geneviève L, Roland L. 1982. General Survey of the carotenoids in Crustacea. In: Britton G, Goodwin T, editors. Carotenoid chemistry and biochemistry. Pergamon: Elsevier. p. 211-224.
Chen SJ, Guo YC, Espe M, Yang F, Fang WP, Wan MG, Tian LX. 2018. Growth performance, haematological parameters, antioxidant status and salinity stress tolerance of juvenile Pacific white shrimp (Litopenaeus vannamei) fed different levels of dietary myo‐inositol. Aquac Nutr. 24 (5): 1527-1539.
Chien YH, Pan C-H, Hunter B. 2003. The resistance to physical stresses by Penaeus monodon juveniles fed diets supplemented with astaxanthin. Aquaculture. 216: 177-191.
Ciji A, Akhtar MS. 2021. Stress management in aquaculture: a review of dietary interventions. Rev Aquacult. 13 (4): 2190-2247.
Díaz AC, Espino ML, Arzoz NS, Velurtas SM, Ponce, NMA, Stortz, CA, Fenucci JL. 2017. Free radical scavenging activity of extracts from seaweeds Macrocystis pyrifera and Undaria pinnatifida: applications as functional food in the diet of prawn Artemesia longinaris. Lat Am J Aquat Res. 45 (1): 104-112.
Díaz AC, Fenucci JL. 2002. Comparative evaluation of different animal protein source in juveniles of Pleoticus muelleri (Crustacea, Penaeoidea). In: Escobar-Briones E, Alvarez F, editors. Modern approaches to the study of Crustacea. New York: Kluwer Academy Plenum Publish. p. 75-78.
Díaz AC, Velurtas SM, Espino ML, Fenucci JL. 2014. Effect of dietary astaxanthin on free radical scavenging capacity and nitrite stress tolerance of postlarvae shrimp, Pleoticus muelleri. J Agr Food Chem. 62: 12326-12331.
Díaz AC, Velurtas SM, Fernandez Gimenez AV, Mendiara SN, Fenucci JL. 2011. Carotenoids from integument, muscle and midgut gland of the red shrimp Pleoticus muelleri (Bate, 1888) (Crustacea, Penaeoidea). Isr J Aquacult-Bamid. 63: 625-631.
Díaz AC, Velurtas SM, Mendiara SN, Fenucci JL. 2013. Correlation between radicals scavenging capacity and carotenoid profile during larval development of Pleoticus muelleri. Invertebr Reprod Dev. 51 (1): 43-48.
Edge R, McGarvey DJ, Truscott, TG. 1997. The carotenoids as anti-oxidants a review. J Photochem Photobio B. 41: 189-200.
Ettefaghdoost M, Haghighi H. 2021. Impact of different dietary lutein levels on growth performance, biochemical and immunophysiological parameters of oriental river prawn (Macrobrachium nipponense). Fish Shellfish Immunol. 115: 86-94. DOI: https://doi.org/10.1016/j.fsi.2021.05.024
[FAO] Food and Agriculture Organization of the United Nations. 2020. El estado mundial de la pesca y la acuicultura 2020. La sostenibilidad en acción. Roma: FAO. 243 p. DOI: https://doi.org/10.4060/ca9229es
Fawzy S, Wang W, Wu M, Yi G, Huang X. 2022. Effects of dietary different canthaxanthin levels on growth performance, antioxidant capacity, biochemical and immune-physiological parameters of white shrimp (Litopenaeus vannamei). Aquaculture. 556: 738276.
Fenucci JL, Pisani E, Díaz AC, Velurtas SM. 2015. Effect of dietary carotenoids on Argentine red shrimp broodstock. J World Aquacult Soc. 46 (1): 57-60.
Focsan AL, Kispert LD. 2017. Radicals formed from proton loss of carotenoid radical cations: a special form of carotenoid neutral radical occurring in photoprotection. J Photochem Photobiol B Biol. 166: 148-157. DOI: https://doi.org/10.1016/j.jphotobiol.2016.11.015
Focsan AL, Polyakov NE, Kispert LD. 2021. Carotenoids: importance idaily life-insight gained from EPR and ENDOR. Appl Magn Reson. 52 (8): 1093-1112.
Gimenez AF, Garcıa-Carreno FL, Del Toro MN, Fenucci, JL. 2002. Digestive proteinases of Artemesia longinaris (Decapoda, Penaeidae) and relationship with molting. Comp Biochem Physiol B Biochem Mol Biol.132 (3): 593-598.
Kirti K, Amita S, Priti S, Jyoti S. 2014. Colorful world of microbes: carotenoids and their applications. Adv Biol. 837891. 13 p.
Kruk J, Szymanska R. 2021. Singlet oxygen oxidation products of carotenoids, fatty acids and phenolic prenyl lipids. J Photochem Photobiol B Biol. 216: 112-148.
Lai TT, Yang CM, Yang CH. 2020. Astaxanthin protects retinal photoreceptor cells against high glucose-induced oxidative stress by induction of antioxidant enzymes via the PI3K/Akt/Nrf2 pathway. Antiox. 9 (8): 729. DOI: https://doi.org/10.3390/antiox9080729
Lenel R, Negre-Sadargues G, Castillo R. 1978. Carotenoid pigments in crustacean. Archs Zool Exp Gen. 119: 297-334.
Lim KC, Yusoff FM, Karim M, Natrah FM. 2023. Carotenoids modulate stress tolerance and immune responses in aquatic animals. Rev Aquac. 15 (2): 872-894.
Lim KC, Yusoff FM, Shariff M, Kamarudin, MS. 2018. Astaxanthin as feed supplement in aquatic animals. Rev Aquac. 10 (3): 738-773.
Marcoval MA, Díaz AC, Espino ML, Arzoz NS, Velurtas MS, Fenucci JL. 2020. Role of dietary photoprotective compounds on the performance of shrimp Pleoticus muelleri under UVR stress. Aquaculture. 515: 734564. DOI: https://doi.org/10.1016/j.aquaculture.2019.734564
Marcoval MA, Pan J, Díaz AC, Fenucci JL. 2021. Dietary bioaccumulation of UV-absorbing compounds, and post-ingestive fitness in larval planktotrophic crustaceans from coastal SW Atlantic. Mar Environ Res. 170, 105433.
Meyers SP. 2000. Papel del carotenoide astaxantina en nutrición de especies acuáticas. In: Civera Cerecedo R, Pérez-Estrada CJ, Ricque-Marie D, Cruz-Suárez L, editors. Avances en Nutrición Acuícola IV. Memorias del IV Simposium Internacional de Nutrición Acuícola La Paz, BCS, México. p. 473-491.
Napoli JL, Horst RL. 1989. Quantitative analyses of naturally occurring retinoid. In: Redfern CP, editor. Retinoid protocols human. New Jersey: Press Totowa. p. 29-41.
Niu J, Li H, Liu Y, Tian J, Chen LX, Huang X, Lin YHZ. 2012. Dietary values of astaxanthin and canthaxanthin in Penaeus monodon in the presence and absence of cholesterol supplementation: effect on growth, nutrient digestibility and tissue carotenoid composition. Br J Nut. 8: 80-91.
Pan CH, Chien YH, Hunter B. 2003. The resistance to ammonia stress of Penaeus monodon Fabricius juvenile fed diets supplemented with astaxanthin. J Exp Mar Biol Ecol. 297 (1): 107-118.
Pereira da Costa D, Campos Miranda-Filho K. 2020. The use of carotenoid pigments as food additives for aquatic organisms and their functional roles. Rev Aquacult. 12: 1567-1578.
Perkampus HH. 1992. UV-VIS Atlas of organic compounds. Part 2. 2nd ed. New York: VCH Wiley. 1525 p.
Pisani E, Díaz AC, Velurtas SM, Fenucci JL. 2014. Effect of dietary carotenoids in the shrimp broodstock Artemesia longinaris. XV Jornadas anuales de la Sociedad Argentina de Biología. Biocell. 38 (2).
Quintana-López A, Hurtado-Oliva MA, Manzano-Sarabia M, López-Peraza DJ, Hernández C, García A, Palacios E. 2019. Effect of rearing conditions on astaxanthin accumulation in the white shrimp Penaeus vannamei (Boone, 1931). Lat Am J Aquat Res. 47 (2): 303-309.
Rahman M, Khosravi S, Chang KH, Lee S. 2016. Effects of dietary inclusion of astaxanthin on growth, muscle pigmentation and antioxidant capacity of juvenile rainbow trout (Oncorhynchus mykiss). Prev Nutr Food Sci. 21 (3): 281-288. DOI: https://doi.org/10.3746/pnf.2016.21.3.281
Roy S. 2000. Strategies for the minimization of UV-induced damage. In: De Mora S., Demers S, Vernet M, editors. The effects of UV radiation in the marine environment. Cambridge: Cambridge University Press. p. 177- 205.
Schiedt K, Bischof S, Glinz E. 1993. Metabolism of carotenoids and in vivo racemization of astaxanthin in the crustacean Penaeus. Meth Enzymol. 214: 148-168.
Stahl W, Sies H. 2003. Antioxidant activity of carotenoids. Mol Aspects Med. 24: 345-351.
Villafañe VE, Sundback K, Figueroa FL, Helbling EW. 2003. Photosynthesis in the aquatic environment as affected by UVR. In: Helbling ER, Zagarese H, editors. UV effects in aquatic organisms and ecosystems. Cambridge: Society of Chemistry. p. 357-397.
Wade NM, Budd A, Irvin S, Glencross BD. 2015. The combined effect of diet, environment and genetics on pigmentation in the giant tiger prawn, Penaeus monodon. Aquaculture. 449: 78-86. DOI: https://doi.org/10.1016/j.aquaculture.2015.01.023
Wade NM, Gabaudan J, Glencross BDA. 2017. Review of carotenoid utilization and function in crustacean aquaculture. Rev Aquacult. 9: 141-156.
Widomska J, Welc R, Gruszecki WI. 2019. The effect of carotenoids on the concentration of singlet oxygen in lipid membranes. BBA-Biomembranes. 1861 (4): 845-851. DOI: https://doi.org/10.1016/j.bbamem.2019.01.012
Yi X, Shen H, Li J, Wei Z, Shentu J, Zhang W, Mai K. 2018. Effects of dietary vitamin E and astaxanthin on growth, skin colour and antioxidative capacity of large yellow croaker Larimichthys crocea. Aquacult Nutr. 24 (1): 472-480. DOI: https://doi.org/10.1111/anu.12580
Zhao X, Wang G, Liu X, Guo D, Chen X, Liu S, Li G. 2022. Dietary supplementation of astaxanthin increased growth, coloration, the capacity of hypoxia and ammonia tolerance of Pacific white shrimp (Litopenaeus vannamei). Aquacult Rep. 23: 101093.
Descargas
Publicado
Número
Sección
Licencia
Derechos de autor 2023 Natalia Soledad Arzoz, M. Alejandra Marcoval, A. Cristina Díaz, M. Laura Espino, Susana M. Velurtas, Jorge L. Fenucci

Esta obra está bajo una licencia internacional Creative Commons Atribución-NoComercial-CompartirIgual 4.0.
Los autores de los artículos publicados en Marine and Fishery Sciences conservan los derechos de autor de sus artículos, a excepción de las imágenes de terceros y otros materiales añadidos por Marine and Fishery Sciences, que están sujetos a los derechos de autor de sus respectivos propietarios. Por lo tanto, los autores son libres de difundir y volver a publicar sus artículos, sujeto a los requisitos de los propietarios de derechos de autor de terceros y sujeto a que la publicación original sea completamente citada. Los visitantes también pueden descargar y reenviar artículos sujetos a los requisitos de citas. La capacidad de copiar, descargar, reenviar o distribuir cualquier material siempre está sujeta a los avisos de derechos de autor que se muestran. Los avisos de copyright deben mostrarse de manera prominente y no pueden borrarse, eliminarse u ocultarse, total o parcialmente. El autoalmacenamiento en servidores y repositorios de preimpresión está permitido para todas las versiones.
Esta revista ofrece a los autores una política de acceso abierto. Los usuarios pueden leer, descargar, copiar, distribuir, imprimir, buscar o vincular los textos completos de los artículos, o usarlos para cualquier otro propósito legal dentro de la licencia Creative Commons 4.0 (BY-NC-SA), sin solicitar permiso previo del editor o del autor. Esto está de acuerdo con la definición BOAI de acceso abierto.






















